
Targeting the
Impossible
TAREXIS discovers novel First-in-Class anticancer targets through real-world patient data — bridging the translational gap with Tx-Finder™ and Tx-Verifier™.

TAREXIS discovers novel First-in-Class anticancer targets through real-world patient data — bridging the translational gap with Tx-Finder™ and Tx-Verifier™.
TAREXIS is a biotech developing innovative therapeutics through novel target discovery. We focus on patients with limited treatment options — those ineligible for targeted therapies and immunotherapies.
We combine real-world patient data, multi-omics analytics, and patient-derived organoid validation to build a high-confidence First-in-Class pipeline across NSCLC, CRC, and PDAC.
Our approach bridges the translational gap: 90% of drugs entering Phase 1 fail. The 3R Framework ensures every target decision is anchored in real-world evidence — before entering expensive clinical development.
Phase 1 failure rate we're engineering around
Target discovery to PDO validation
All pipeline assets, no same-target ADC competition

The Translational Gap ("Valley of Death") — bench-to-bedside translation fails for 75–90% of preclinical results. TAREXIS's 3R Framework is designed to bridge this gap.
We advance drug development on the 3R framework — ensuring every target reflects real unmet medical need combined with high drug potential and commercial viability.
Needed in real clinical practice and the market?
Expressed in real patients?
Effective in real patients?
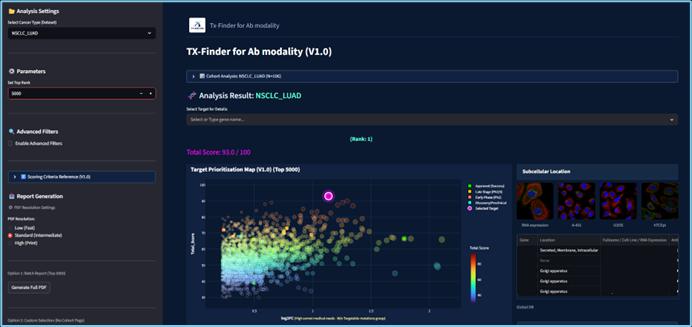
Two integrated platforms systematically narrow down high-confidence targets and pre-validate clinical performance — before IND-enabling studies begin.
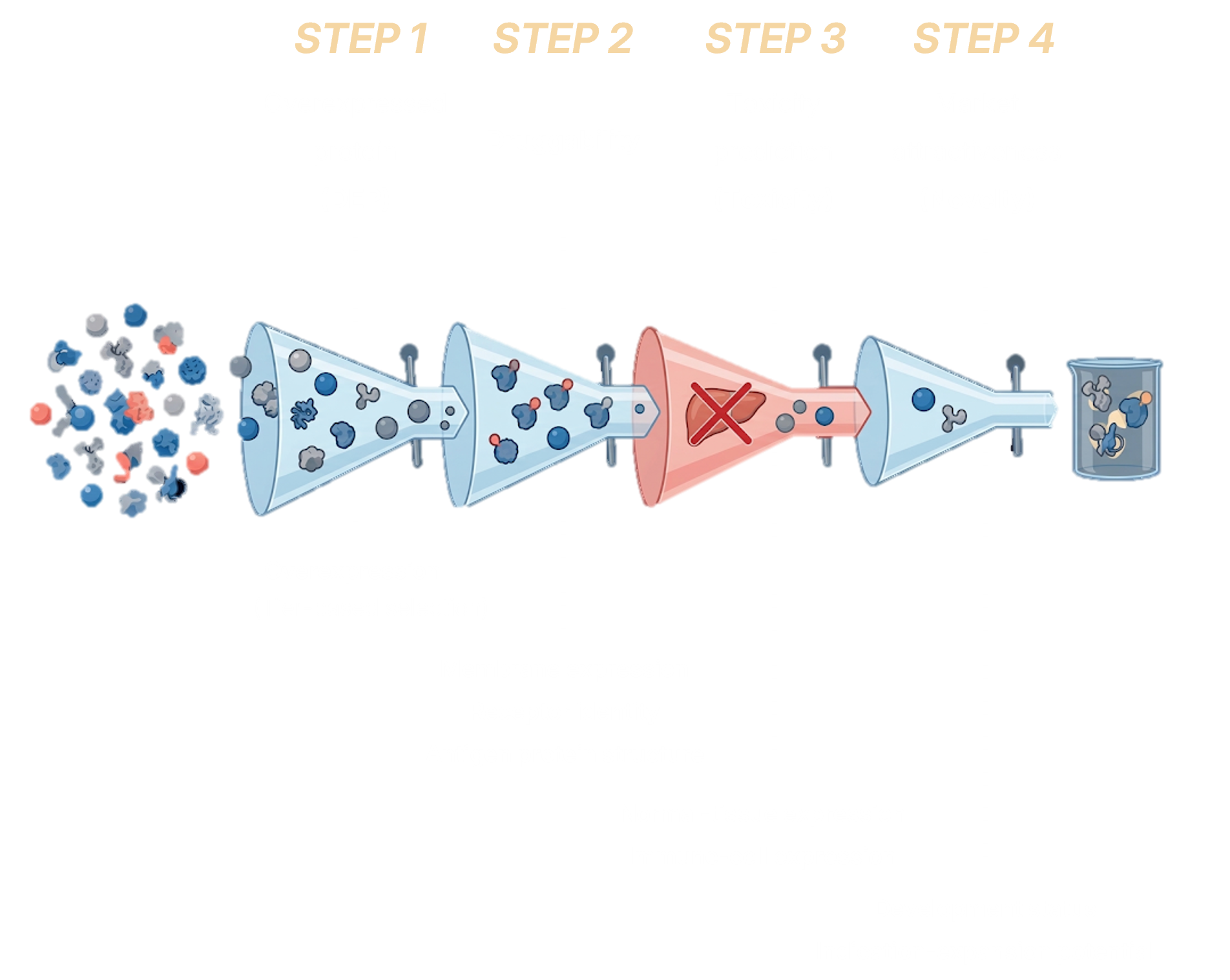
A 4-step novel-target scoring system integrating RWU and RWD to auto-prioritize high-confidence FIC candidates from 10,000+ starting points. Integrates AlphaFold DB, HPA, GTEx, ClinicalTrials, and in-house omics.
4-Step Candidate Filtering Process
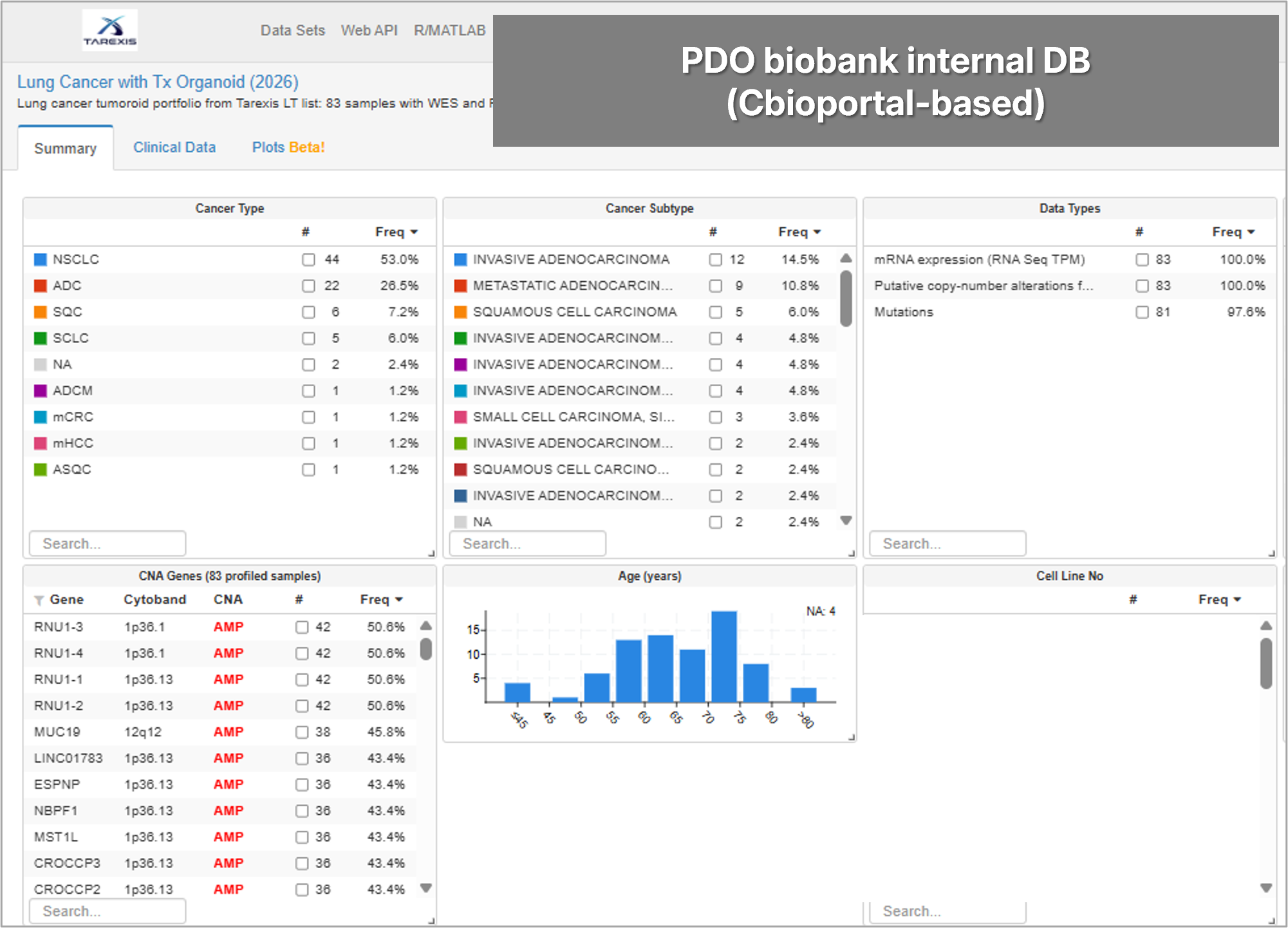
Early efficacy validation using PDOs cultured directly from real patient tumor tissue. Captures tumor heterogeneity to pre-validate clinical performance before IND-enabling studies — minimizing late-stage failure risk.
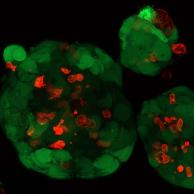
Patient-derived organoids (PDO) — real tumor tissue from TAREXIS's PDO biobank
All assets are First-in-Class with no competing same-target ADC programs — targeting patients ineligible for existing therapies across multiple solid tumor indications.
| Program | Class | Indication | Modality | Stage | Partner |
|---|---|---|---|---|---|
| ▸ Novel Target Pipeline | |||||
| TX-101 | FIC | LUAD · LSCC | ADC/BsADC | Partner B (China) | |
| TX-102 | FIC | LUAD · LSCC | ADC/BsADC | Partner B (China) | |
| TX-103 | FIC | LSCC | ADC/BsADC | Partner B (China) | |
| TX-104 | FIC | LUAD · LSCC | ADC/BsAb/BsADC | Partner M (France) | |
| TX-201 | FIC | CRC | ADC | — | |
| TX-301 | FIC | PDAC | ADC | — | |
| ▸ Bispecific Antibody Platform | |||||
| TX-BSU | FIC | Universal | BsAb/BsADC | — | |
LUAD: Lung Adenocarcinoma · LSCC: Lung Squamous Cell Carcinoma · CRC: Colorectal Cancer · PDAC: Pancreatic Ductal Adenocarcinoma
3R Framework + Tx-Finder™ + Tx-Verifier™ = higher clinical success probability, faster development, and stronger commercial outcomes.
Unmet need → target → patient-derived sample validation. 3R Framework achieves higher clinical hit rate.
Target discovery → PDO validation in ~12 months. Real-time analytics + PDO-based early PoC.
Weighs efficacy, safety, drug value, and market potential jointly at discovery — higher L/O probability.
Applies across ADC, BsAb, TCE, and PDC. Modality fit pre-screened: 1 target → multiple modalities.
The global NSCLC market is on a 9% CAGR trajectory with a large patient population still unserved by current targeted and immune therapies.
From $34.8B in 2023 — CAGR 9%. ~18% of total oncology market.
Source: Evaluate Pharma 2024
Of total NSCLC are cold tumors ineligible for immunotherapy. LSCC: 95.5% WOT. LUAD: 55% WOT.
Massive underserved patient population
Enhertu ($6.5B), Trodelvy ($2.8B), Datroway ($1.5B) validate FIC ADC value in NSCLC.
Analyst consensus 2028E
Interested in partnership, co-development, or licensing? We welcome conversations with investors, pharma partners, and research collaborators.
Tell us about your interest and we'll be in touch.